|
Going to test my students in our O-Level Pure Chemistry Tuition Classes on the same question and see how they fare for it □ PS: Hmm, just realised this is quite a tricky question. You can refer to Kinetic Particle Theory to learn more on 3 states of matter. It could also mean that simple molecular structures are packed closely together in a regular pattern when they are in solid state. DO NOT be tricked to think that iodine is an ionic compound due to the word “crystals” which is usually associated with ionic compound. At room temperature, iodine is a solid so that diatomic molecules are packed very closely together in a regular pattern, to form “crystals”. Option D: Iodine is an element and it exists as a diatomic molecule (i.e. Option C: Carbon dioxide exists as simple molecular structure (i.e. Option B: Graphite is an element BUT it exists as giant molecular structure of carbon atoms held together by strong covalent bonds. Option A: Copper is an element BUT it exists as positive metal ions surrounded by ‘sea of delocalised valence electrons’. (ii) also forms crystals composed of small molecules? Strong electrostatic forces of attraction holds the positive ions and the negative ions together in the giant ionic/crystal lattice structure.Īs such, a large amount of energy is required to overcome these strong forces. They tend to exist as solids at room temperature and pressure. Ionic compounds tend to have high melting point and boiling point. Which of the following is most likely to be the melting point of an ionic solid? Today, we shall look at a few more exam-based Chemistry MCQ questions on this topic: In the last blog post, we went through a common exam-based question on Chemical Bondings and revised briefly on: I will be teaching a committed group of students to truly appreciate the Key Essential Concepts and the Keywords associated with it. I will be conducting a 1-Day Chemical Bonding Intensive Revision Workshop on Wednesday, 4th June during the School Holiday. 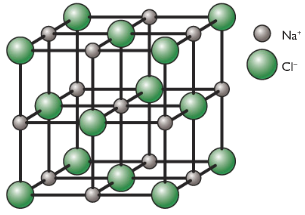
Feel free to “share” this post with anyone who might benefit from it.įor those that need help to sort out this essential topic quickly and confidently, i have good news for you. Many students are not aware that there are Two Types of Bonds/Forces when it comes to carbon dioxide which is a SIMPLE MOLECULAR STRUCTURE, which are:ġ) Strong Covalent Bonds between the atoms WITHIN the small moleculesĢ) Weak Intermolecular Forces of Attraction BETWEEN the small molecules 
Small amount of energy is required to overcome it.” This is so much better, with the correct keywords all given to the examiner. “Weak intermolecular forces of attraction exists between the small covalent molecules of carbon dioxide. 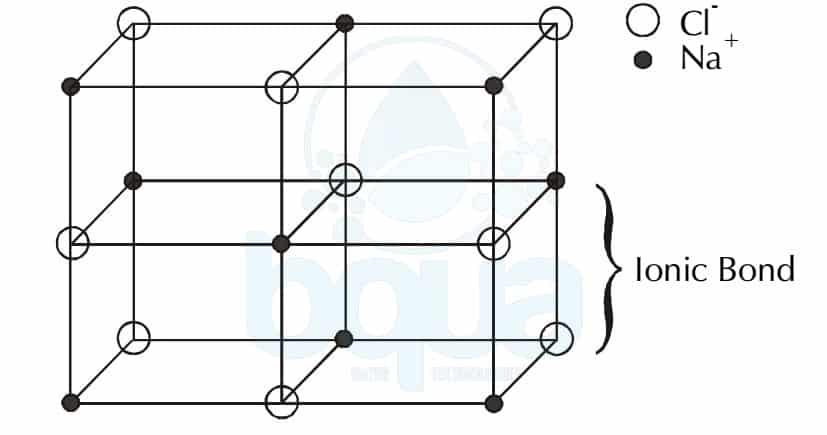
“The covalent bonds in the carbon dioxide molecule is weak and thus requires small amount of energy to overcome it.” which is WRONG! It only shows the examiner that you are weak in your concepts. Many students would mention the following: It is always a Common Mistake!Įxplain why carbon dioxide is a gas at room temperature and pressure. Some also do not understand the difference between “TYPES OF BONDING” and “TYPES OF STRUCTURES”ĭo note that whenever the question ask for “PROPERTIES” of a substance, we are really asking you to link the physical properties of a substance to the “TYPES OF STRUCTURES” and NOT “Types of Bonds”. They often USE THE WRONG KEYWORDS or have a LACK OF KEYWORDS such that their teacher have no choice but to minus marks. They would tell you that they are pretty good with this topic but whenever you analyse their exam papers, you will realise that they don’t really know the topic well. However, many do not realise the importance of this topic and take it very lightly. there are so many topics that requires students to have a strong foundation of Chemical Bondings that it will take a lot of typings for me to list them down one-by-one. They never realised that mastery of this topic would have helped them to truly understand they key concepts in most follow up topics in Secondary 3 and 4 such as:Ĥ) Qualitative Analysis (Identification of Ions)Įtc…. Many students neglect this very fundamental yet important topic in Secondary 3 when they learn Chemistry from their school teachers. Tips for GCE O-Level Chemistry: CHEMICAL BONDING
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
